Human Genome Editing Workshop with the Swiss life sciences community
Workshop summary
On February 18th 2021, the Forum for Genetic Research of the SCNAT hosted at the LS2 Annual Meeting an interactive online workshop to explore different aspects of human genome editing together with members of the Swiss life sciences community.

Acceptability, hopes and concerns
In his input, Prof. Jacob Corn from the ETH Zürich explained that genome editing technology has been around since the 1990s, but has recently received an enormous boost due to the discovery of the CRISPR system. The high flexibility of the CRISPR system makes it a tool that can be relatively quickly and easily deployed. This leads to a democratization of research and rapid progress, but at the same time also raises important questions regarding ethics and regulations as the tool becomes more distributed. Somatic editing of adult cells can be viewed similarly to a medicine, where for example bone marrow cells of a patient are edited to cure a disease. Germline editing of sperms, eggs or zygotes on the other hand leads to changes that will be passed down to the next generation. It is this potential application of genome editing that caused a lot of worries and discussions with regards to unintended consequences and misuse. However, germline editing can also be used for fundamental research such as the investigation of causes of human infertility or miscarriages.
In the live-polling (see link to full results below) our audience expressed a high acceptance of somatic genome editing for research and therapy. Accordingly, curing genetic diseases as well as cancer were the biggest hopes associated with somatic genome editing. To this, Jacob Corn added that somatic genome editing is at the moment primarily researched for comparatively easy targets such as blood disorders or eye diseases. However, some of the most devastating disorders including progressive neurological disorders like Huntington's disease are extremely difficult to target and edit. While CRISPR holds many promises, we are still at the very beginning of understanding and applying this technology more broadly to human diseases and much more research and development are needed.
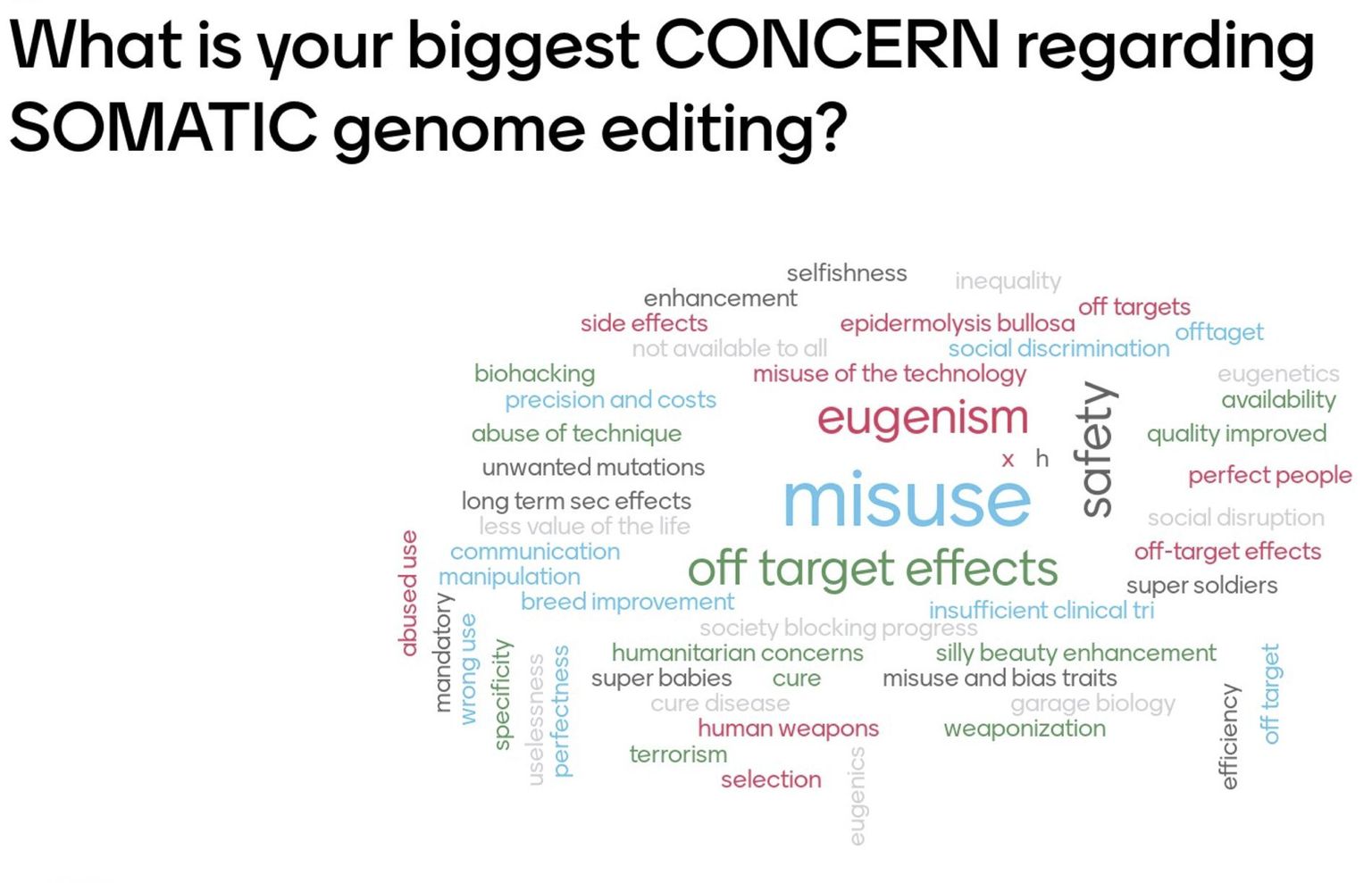
As major concerns, the audience named safety issues such as unintended off-target effects but also the intentional misuse e.g. for military purposes or eugenics.
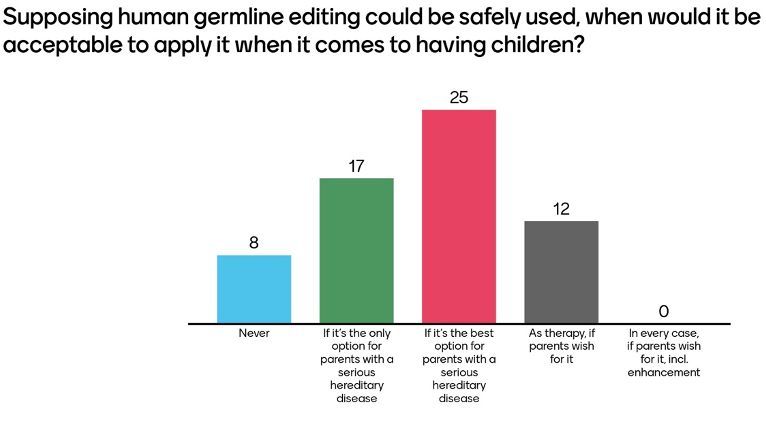
Not surprisingly, editing in the human germline was on average seen more critically than somatic editing and opinions varied more widely. For instance, about 13% of respondents (8 out of 62) thought that human germline editing should never be applied while 20% (12 out of 62) would leave the decision up to the parents as long as it is for therapeutic purposes. In this context, Jacob Corn stressed that the purpose of research into therapeutic genome editing should be to provide patients or parents with options and tools but that it will always have to be their own choice whether they want to make use of such a tool or not.
Finally, for both somatic and germline editing, applications for enhancement, i.e. to introduce improved or new characteristics, were considered largely inacceptable by our participants. However, Jacob Corn pointed out that there are also more surprising cases of somatic enhancement that are being discussed such as introducing tolerance for microgravity, potentially allowing humans to travel further in space and reaching new frontiers.
Governance and responsible research
In his input, Prof. Hervé Chneiweiss from the Ecole des Neurosciences in Paris and member of the WHO expert committee on human genome editing gave us an overview of currently ongoing international discussions on legal, societal and ethical aspects as well as governance options for human genome editing. The WHO advisory committee put forward three guiding principles:
- transparency, expressed in the decision to develop a registry for planned and ongoing research and development projects
- responsible stewardship of science, resulting in a WHO statement that at the current time it would be irresponsible to proceed with clinical research on human germline editing
- inclusivity, for instance by sharing information with a lay audience
There are two types of issues that have been considered: procedural issues and values. As for procedural issues, clarity, transparency and accountability are essential. Science must be open so that decisions can be based on the best available scientific evidence. In addition, we need approved policies for human genome editing as a basis for trust. Laws and regulations should be promoted in countries where there are currently little or no regulations. Also important is program management and research training as well as public awareness raising. For research, we must assure high quality but also ethical practices in order to maximize the potential benefit and minimize the potential harm. For this, we need a mobilization of the scientific community but also responsible stewardship of research resources.
Regarding substantive issues, we will need inclusiveness, not only of scientists but also different cultural, social and religious communities, fairness, social justice and non-discrimination, equal moral worth of all people including people with genetic disabilities, solidarity and a commitment to live and work in harmony and to make human genome editing a shared benefit as well as global health equity.
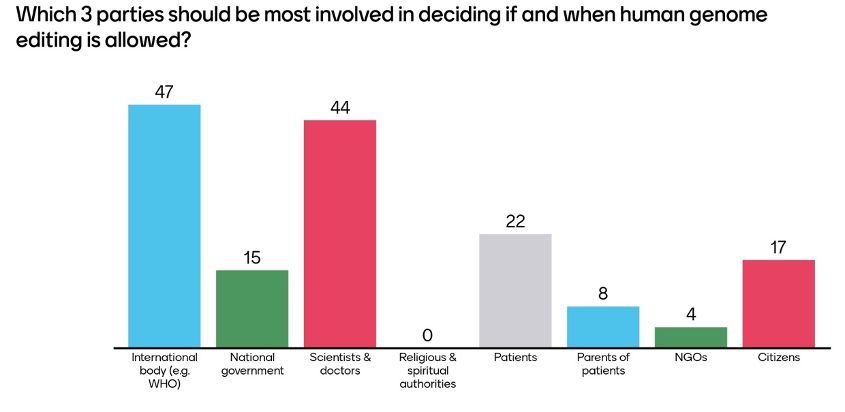
In the live polling, the audience considered international bodies as well as scientists and doctors as the most important actors to include in the governance of human genome editing. While Hervé Chneiweiss considered the apparent trust in these two groups as positive, he also stressed the importance of inclusiveness and in particular the involvement of citizens but also of religious and spiritual leaders in the decision process.
When asked about important elements of responsible research, the participants most often named transparency and related topics such as communication and open science. Further, personal integrity and honesty, ethics, safety and risk-benefit evaluations and reproducibility were proposed. Interestingly, about half the respondents thought that questions of responsible research should be discussed and addressed more often in their research group.
Finally, the participants made suggestions on how a real dialogue between scientists and society on human genome editing could be fostered. Many proposals centred around attractive, honest and respectful science communication in a large variety of formats including open public fora, movies and radio shows and addressing citizens of all ages.
The full results of the live polling can be found as a PDF below.
-----
This workshop was organized by the Forum for Genetic Research of the Swiss Academy of Sciences (SCNAT) together with Life Sciences Switzerland (LS2).
- Host: Greta Guarda (Istituto di Ricerca in Biomedicina, Università della Svizzera italiana)
- Moderator: Anna-Katharina Ehlert (reatch)
- Speakers: Jacob Corn (ETH Zürich) and Hervé Chneiweiss (Ecole des Neurosciences, Paris Ile de France).
- Organizing committee: Martine Jotterand (Forum for Genetic Research), Greta Guarda, Michaël Bachmann (University of Geneva) and Bernard Baertschi (University of Geneva).
The workshop took place as a Zoom webinar, live polling was conducted using Mentimeter.